
Above all, the electron escape depth (measurement depth) can be changed by tilting the sample with respect to the analyzer. Therefore, it is likely that the powder is a mixture of CuSO 4 and Cu (NO 3) 2. Collect principal Cu LMM peak, as well as Cu2p. such as lineshape, peak energy and peak intensity for Cu2p, Cu(L,M.,M,). The high-resolution data shows that Cu exists as Cu 2+, S as sulfate, and N as nitrate. Copper Transition Metal Chemical state differentiation can be difficult with XPS only. Oxidized surfaces Washington State University-Pullman, WA XPS Sp Cr 2p / 7 r r k k Cr 2p -data x 10 3 4 6 8 10 12 S 590 580 570 Binding Energy (eV) XPS Sp Cr 2p / 2. Satellites are also seen on the Cu 2p spectra. The oxide layer resulted in extra peaks (shoulder at higher BEleft of the main line). These parameters are very useful for identification of the different states present in the surface but they are difficult to quantify as relative amounts of each species. Diagnostic spectra for XPS analysis of Cu-O-S-H compounds. Oxidized and clean Cu 2p spectra (right). Poulston et al., in their study of surface oxidation and reduction of Cu 2O and CuO, have used both the Cu LMM and the Auger parameter to distinguish Cu(0), Cu(I) and Cu(II). They also note the distinctive Cu L 3M 4,5M 4,5 peak at 916.5 eV for Cu 2O. have shown (in their Figure 8) the distinctly different peak shapes of the X-ray generated Auger LMM spectra for copper as the metal, Cu 2S and CuS.

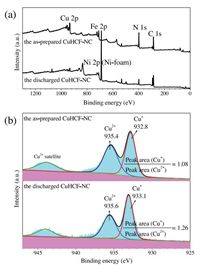
The use of the modified Auger parameter (2 p 3/2, L 3M 45M 45) as well as an inspection of the Auger peak-shape do allow for a more accurate assignment for these species and has been used effectively. In Figure 3, we see high energy resolution spectra acquired from the copper samples, A. The main peaks occur at kinetic energies of ca. The ability to detect and quantify this shift makes XPS such a powerful analytical technique. The diagram below shows a real XPS spectrum obtained from a Pd metal sample using Mg Ka radiation. Of note here is the statistically similar BE values for the Cu metal and Cu(I) oxide species. peaks in finer detail, then we see that even for the same element, the peak energy may shift depending on the surface chemistry. Table 1 lists Cu 2 p 3/2 BE and modified Auger parameter values from a survey of literature sources compiled in the NIST Database.
